AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Sas tutorial for clinical data management11/24/2023  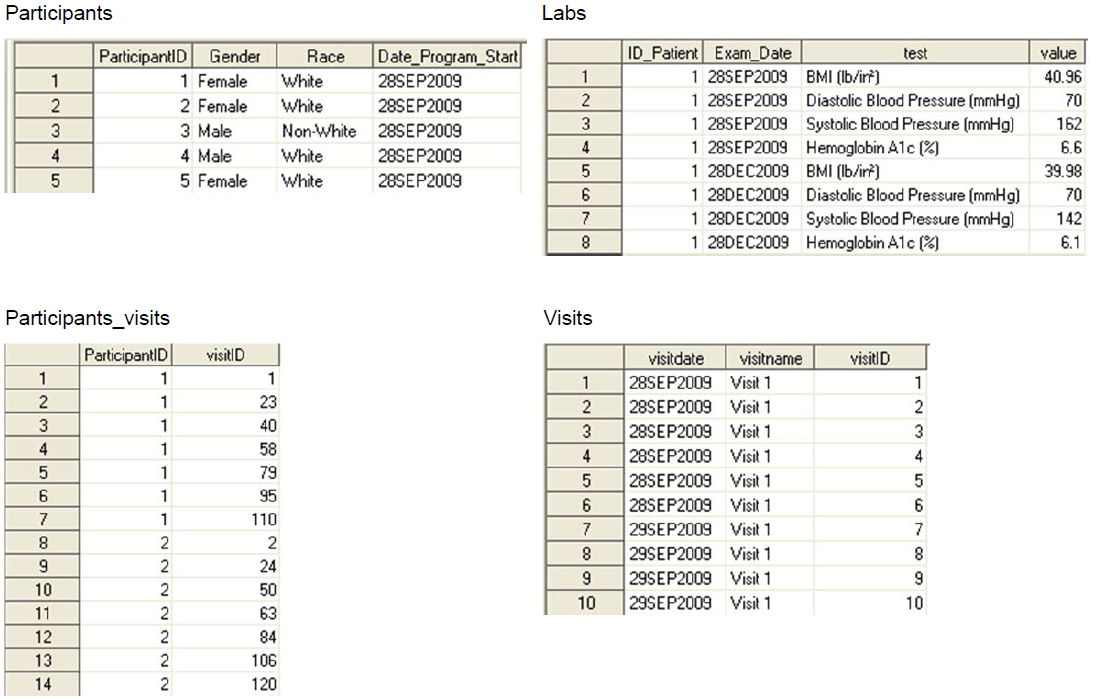
SAS programming skills are necessary for conducting statistical analyses and generating clinical study reports.Ĭlinical trial design: This involves designing clinical trials, including sample size calculation, randomization, and blinding.Ĭlinical trial monitoring: This involves monitoring clinical trial data and ensuring data quality and protocol compliance. 
SAS programming: This involves using SAS programming language for data manipulation, analysis, and reporting. 
SAS supports CDISC standards, including the Study Data Tabulation Model (SDTM) and Analysis Data Model (ADaM). Statistical analysis: This involves performing statistical analyses on the clinical trial data, such as hypothesis testing, regression analysis, survival analysis, and ANOVA.Ĭlinical trial reporting: This involves generating tables, listings, and figures for clinical study reports and regulatory submissions.ĬDISC standards: Clinical Data Interchange Standards Consortium (CDISC) standards are used for data exchange and submission to regulatory authorities. There are also various SAS certification programs available for professionals seeking to enhance their SAS programming skills and knowledge.Ĭommon Topics Related to Clinical SAS Tutorials:ĭata management: This includes data cleaning, quality control, and data integration from various sources. Proficiency in Clinical SAS is a highly sought-after skill in the pharmaceutical and clinical research industries, and many companies require SAS programming experience for roles such as clinical data managers, biostatisticians, and statistical programmers. It also includes a variety of statistical procedures, such as regression analysis, survival analysis, and analysis of variance (ANOVA). Key features of Clinical SAS include the ability to handle large and complex datasets, automate data management tasks, perform advanced statistical analyses, and generate tables, listings, and graphs for clinical trial reports. It is also used for regulatory submissions, where the output from SAS programs is used to support drug or medical device approval. It is a powerful tool that provides a wide range of functionalities, including data manipulation, transformation, and analysis, as well as data visualization and reporting.Ĭlinical SAS is used in various phases of clinical research, including study design, data collection, data analysis, and reporting. Clinical SAS (Statistical Analysis System) is a software suite commonly used in the pharmaceutical and clinical research industries for data management, statistical analysis, and reporting of clinical trial data. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
